Dear community,
From Dr. Federici 's lab we wanted to share with you some good news about a project we are working on with collaborators in Valdivia (Dr. Guillermo Valenzuela), who can immunize alpacas against a variety of pathogens to produce nanobodies. We are implementing an open-source, golden-gate-based platform to streamline nanobody cloning, screening and functionalization for downstream applications.
The pipeline consists in i) alpaca immunization; ii) nanobody screening by bacterial display; iii) cloning of successful nanobodies using only golden-gate enzymes and Reclone/uLoop syntaxis, therefore allowing a universal and modular solution to clone virtually any nanobody from the library (no need of PCR primers, nor custom restriction enzymes design), iv) functionalization of nanobodies through golden-gate assembly in pTI plasmid for downstream applications.
So far, we can report the following progress:
- We have modularized nanobodies anti-eGFP and anti-SARS-CoV-2, which sequences (between C and D) are available as levels 0 in pL0R at:
-
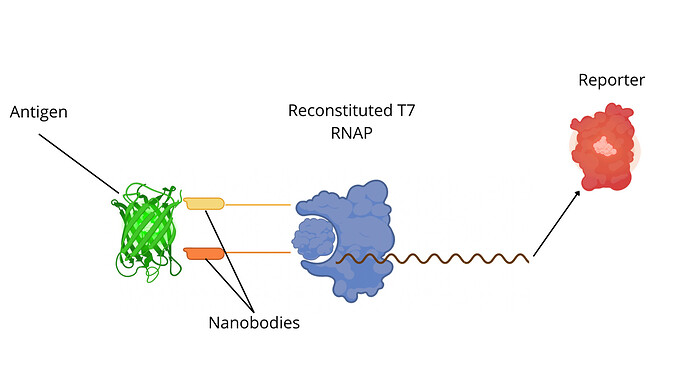
Thanks to a collaboration with Dr. Styczynski (Georgia Institute of Technology), we have domesticated, modularized and reassembled in pTI the TLISA system (a split T7-based nanobody biosensor Mc Sweeney et al, 2025). Each individual level 0 part can be found in our Github repo. In a few words, the N-terminal and C-terminal fragments of the split T7 RNA Polymerase are each fused to a nanobody. Upon the non-competitive interaction of these nanobodies with a target, the C-term and N-term fragments reconstitute into a fully functional T7 RNAP, triggering the expression of a pT7-reporter (Fig.1, Top). Credits to @vferrando and @tfmatuteuc-cl for the titanic work of building the libraries of all the level 0 parts.
-
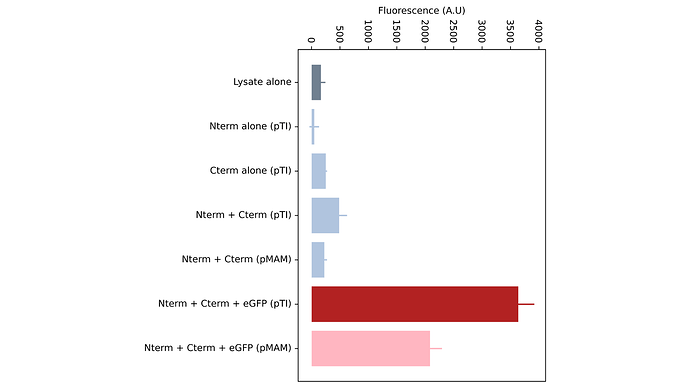
We have validated that the split T7-system is working fine in pTI once modularized, using the anti-eGFP nanobodies as a proof-of-concept. Below is a figure with the results we obtained, comparing the performance of the domesticated system (pTI) against the original vectors (pMAM) in home-made cell-free reactions. (Fig.1, Bottom).
We have modularised other parts such as periplasmic tag, exportation peptides, linkers, fusion domains. This project is still in progress. We are currently working on split systems and immunizing against local pathogens of interest to validate these tools for “immunisation-to-functionalization”. Anyone interested in this project is invited to raise a hand and join.
Fig.1 - Top: Principle of the split-T7 nanobody-based biosensor assay. Bottom: Endpoint fluorescence of the pT7 reporter (here, mScarlet). The domesticated system in the pTI vector (dark red) is compared to the original biosensing system in pMAM vector (light pink). Other colors are the different negative controls required for the assay validation.