Hi All
This is probably not news to protein expression nerds but I was looking for a reference for IMAC purification of low abundance proteins and learned something that I hadn’t seen data for before.
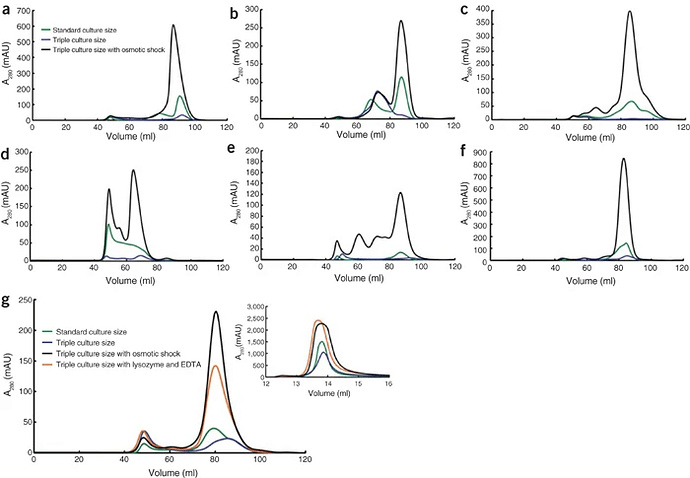
We had some low abundance proteins that weren’t giving good purity from IMAC purification because endogenous protein was binding, which we’ve now resolved by changing expression vector. Fundamentally, you should initially focus on getting high expression yields because the competition effect with endogenous protein is real for a given fixed volume of lysate with low vs high expression levels of your target protein BUT what I learned from this paper was that high volumes of lysate strip the column due to metal chelators. So if your approach to low expression is to scale up culture size, you can actually get worse yields as you try to put more volume through the column and they have some nice data to show this.
If you’ve exhausted options to increase expression that don’t involve scaling up expression volume and you’re finding that you’re not getting good yields from your IMAC columns, consider following the protocols in this paper!
Magnusdottir, Audur, et al. “Enabling IMAC purification of low abundance recombinant proteins from E. coli lysates.” Nature methods 6.7 (2009): 477-478.
the underlying cause for the reduced target protein binding is not the result of native E. coli proteins competing with the His-tagged protein for the immobilized nickel-ion binding sites. We determined the amount of nickel present on the different columns before and after sample load and found that the decrease in binding capacity correlated with loss of immobilized nickel ions from the column
IMAC is very sensitive to the presence of metal chelators1, and the E. coli lysate contains many unspecific weak chelators such as dicarboxylic acids from the citric acid cycle. Under stress conditions, E. coli can also produce highly specific metal chelators, metallophores5. We speculated that such metallophores, if produced, would be mainly associated with the periplasmic space of E. coli but not with the cytosol… By removing the periplasmic material before cell lysis [JM note: via osmotic shock] and loading the lysate on IMAC columns, we observed a tenfold increase in yield of His6-GFP when it was diluted with E. coli lysate before purification to simulate a low-abundance protein
Although a considerable fraction of recalcitrant proteins will still need customized production, our findings show that it will be possible to increase the yield for many His-tagged proteins and enable IMAC purification for very low-abundance proteins. We believe that our method could greatly increase the number of recombinant proteins available for biochemical investigations. In addition, by removing the periplasmic material, a substantial scale-down of the whole purification setup, including column sizes and buffer volumes and so forth, may be possible without any loss of final yield or purity.